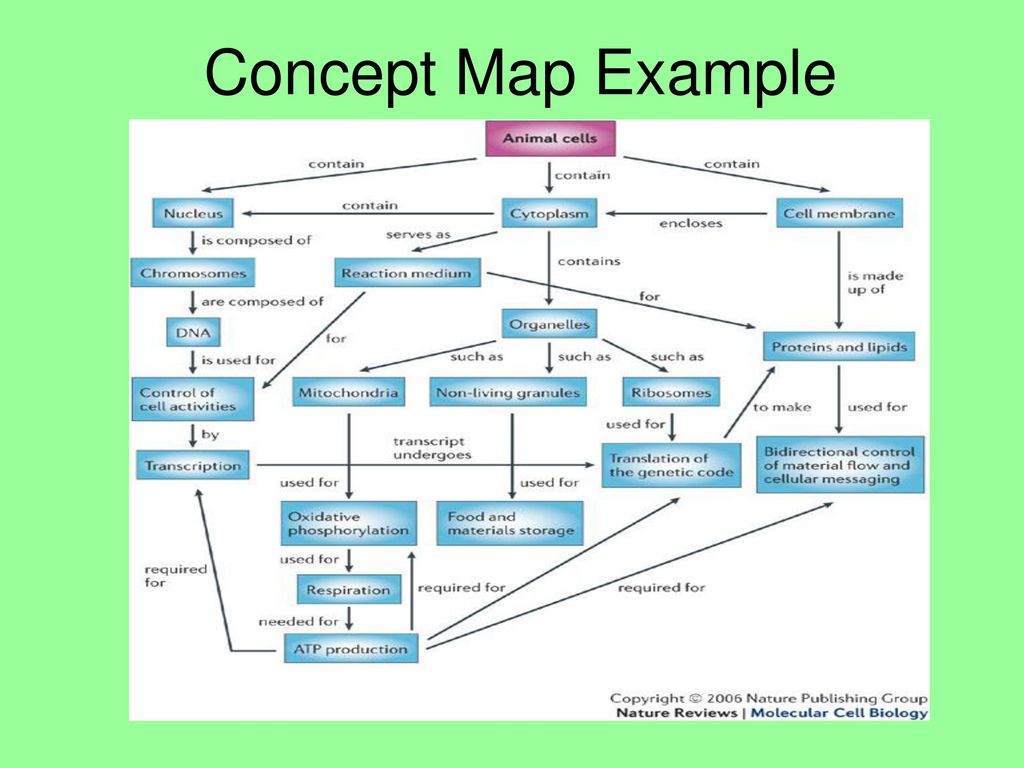
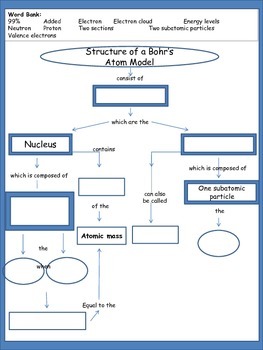
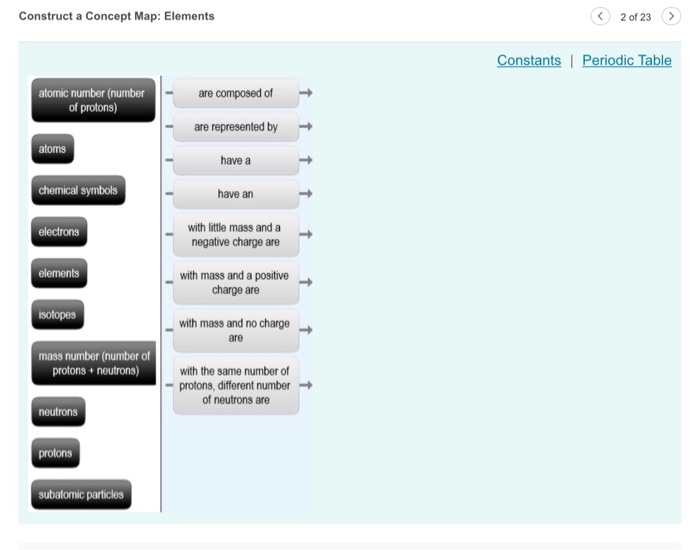
Atoms Have Subatomic Particles Concept Map
The positively charged protons weigh about 1 836 times more than electrons.
Atoms have subatomic particles concept map. Examines the particles that make up matter such as subatomic particles atoms and molecules. Please update your bookmarks accordingly. For example a piece of aluminum foil is shiny silver in color. Germanium cobalt or mn 57.
Displaying all worksheets related to subatomic particles. Please check your answers and forward any problems to mrs. What differentiates one atom from another is the relationship they establish between them. Determine the number of protons and electrons in an element.
Which element has 19 electrons. Countthe numberof protons neutrons and electrons in the bag. Write a z format for the species in question 5. An atom is defined as the smallest unit of an element that still has the properties of that element.
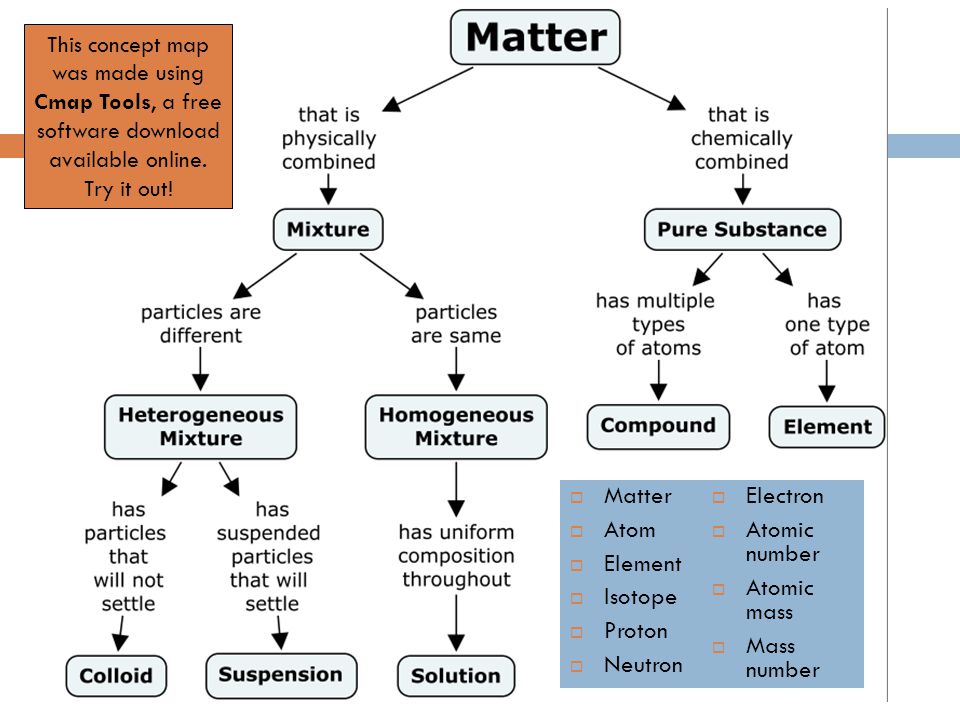
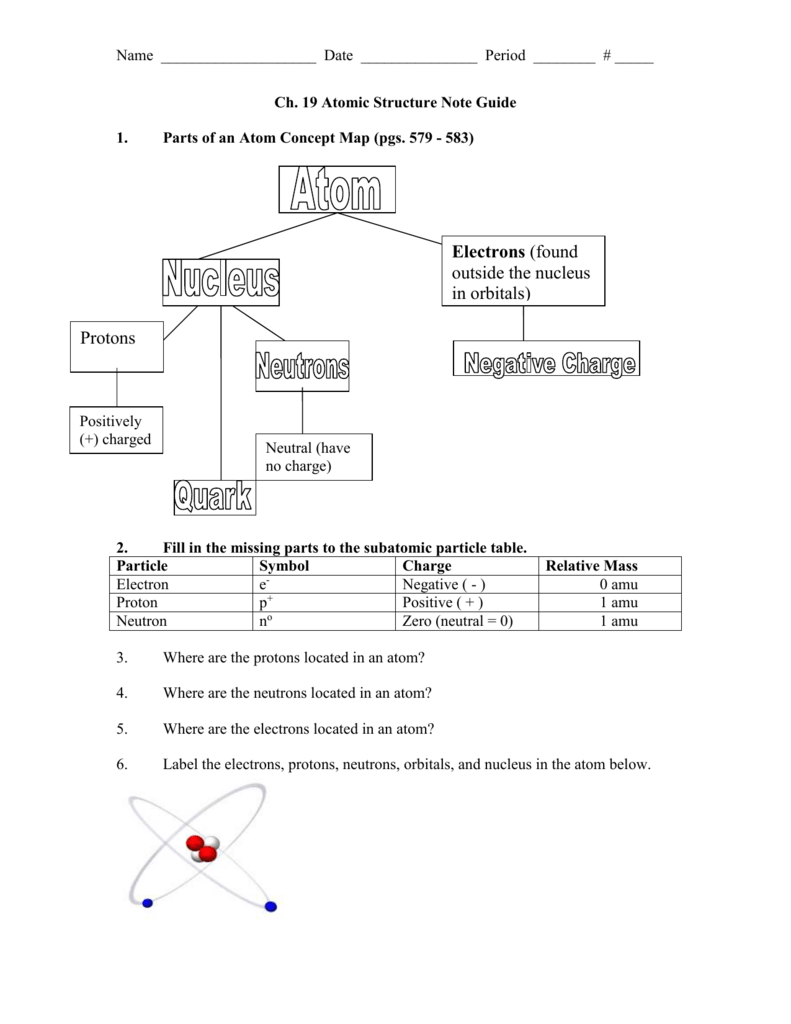
Which element s has have 32 neutrons. The negatively charged electrons are the lightest subatomic particles. The number of protons is called. Protons neutrons and electrons are the subatomic particles that form the structure of the atom.
Calculate the subatomic particles for zn 67 radium the aluminum cation mg 23 and. This page will be removed in future. Which natural isotope of platinum is most abundant. A fundamental characteristic of each element is that the atoms of that element all have the same number of protons.
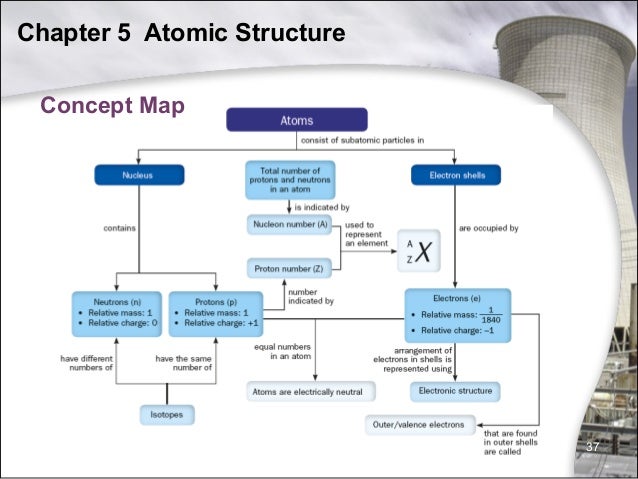
Describe the locations charges and relative masses of protons and electrons. Subatomic particles include electrons which are the negatively charged almost massless particles which nevertheless account for most of the size of the atom. It includes the heavier building blocks of the small yet dense nucleus of the atom the positively charged protons and the electrically neutral neutrons. What do you notice about the relative mass of the proton and neutron.
Subatomic particles relative mass atomic mass units particle symbol proton neutron o electron relative charge 1 0 1 absolute mass kg 1 673 x 10 kg 1 675 x 10 kg 9 109 x 1031 kg location in the atom nucleus nucleus electron cloud 1 0 1. Save as pdf page id 213160. The field of subatomic particles has expanded vastly with the construction of powerful particle accelerators for studying the high energy collisions of electrons protons and the. Atoms and subatomic particles last updated.
What about the electron. Some of the worksheets for this concept are atomic structure work ion symbol protons electrons charge answer key atoms and their parts subatomic particles atoms and isotopes work subatomic heavyweights work answers name period date subatomic particle counting subatomic particles work answers. Progress practice now. We have moved all content for this concept to for better organization.
This indicates how strong in your memory this concept is.